Research
NCCR AntiResist is transforming antibiotic research and discovery by developing laboratory models that replicate physiological interactions between bacteria and human tissue based on conditions observed in patient samples. This innovative in-vitro modelling of in-vivo conditions provides novel opportunities for antibiotic discovery and gives new hope in the fight against antibiotic-resistant infections.
Scroll down for an overview of our research or dive right in with the links below.
Why is our research necessary?
Because bacteria rapidly evolve, they are able to develop resistance to antibiotics. The ability of bacteria to deflect the effects of antibiotics has become a global medical threat. After thirty years of stagnation in the development of new antibiotics, we are faced with the very near prospect of a post-antibiotic era entailing a massive health and economic burden.
While new, conventionally discovered antibiotics provide a temporary reprieve, AntiResist strives to develop more sustainable and global approaches to address the antibiotic innovation gap. With a fundamental shift towards in vivo (inside the living organism) antibiotic research, we are providing the standard for continual antibiotic discovery to keep pace with resistance evolution for generations to come.

How is our research different?
Our research represents a fundamental shift in how antimicrobial compounds are discovered and tested.
Conventionally, antibacterial drug research and testing is mostly done using standardised laboratory culture conditions, which do not accurately imitate the real conditions bacteria face inside the human patient (a).
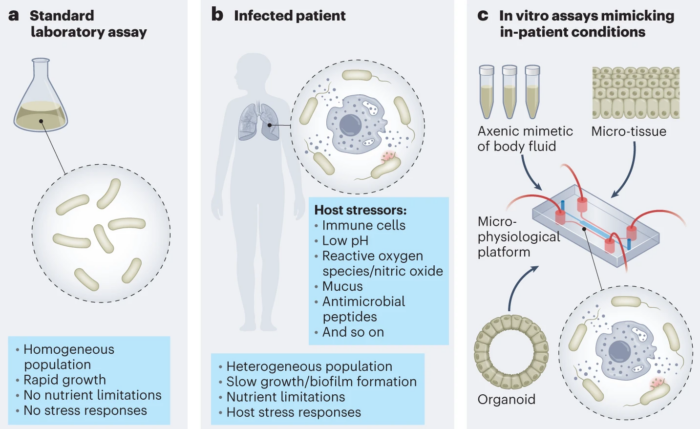 How are conventional research approaches limited? The internal environment of the human body is physically, chemically and biologically complex, creating various stresses for bacteria through nutritional limitations or host defence mechanisms. This highly adaptive host environment is shaped by diverse human cells and biomolecules (b) which are not considered by conventional antibiotic development approaches. An example of how a laboratory culture does not reflect the human host comes from genetics, which has identified bacterial genes that are inactive in a laboratory-cultured environment, but which are active when bacteria are infecting human tissue and support them to thrive. This reveals a substantial blind spot in antibiotic research, and there could be many more. Without an accurate view of how bacteria interact with the human body, scientists struggle to identify pertinent bacterial vulnerabilities and avenues of possible antibacterial action.
How are conventional research approaches limited? The internal environment of the human body is physically, chemically and biologically complex, creating various stresses for bacteria through nutritional limitations or host defence mechanisms. This highly adaptive host environment is shaped by diverse human cells and biomolecules (b) which are not considered by conventional antibiotic development approaches. An example of how a laboratory culture does not reflect the human host comes from genetics, which has identified bacterial genes that are inactive in a laboratory-cultured environment, but which are active when bacteria are infecting human tissue and support them to thrive. This reveals a substantial blind spot in antibiotic research, and there could be many more. Without an accurate view of how bacteria interact with the human body, scientists struggle to identify pertinent bacterial vulnerabilities and avenues of possible antibacterial action.
How do we address this gap? Identifying and exploiting the possibilities offered by the human host environment is central to our approach at NCCR AntiResist. We start by taking tissue samples from infected patients, and study them closely. We then replicate this in-vivo infection environment in vitro (in the laboratory) by building axenic, microtissue and organoid models to study infection and treatment (c).
Accurately modelling in-patient infection conditions in vitro creates a realistic physiological environment to probe in vivo states, which can be observed using high resolution microscopy and other methods to elucidate the dynamics of the infection process, as well as the effectiveness of anti-infective compounds. We believe that this innovative strategy to replicate the environment of the human body will fundamentally change infection research and help ensure infection control for future generations by revitalising the antibiotic discovery pipeline.
For further details about our research approach, see 2024 Comment by Sollier et al. in Nature Microbiology.
Interdisciplinary approach
The NCCR AntiResist research team includes experts in microbiology, biochemistry, human biology, pharmacology, bioengineering, and clinical medicine across multiple institutions in Switzerland and abroad, who work collaboratively to identify new antibiotics and antimicrobial strategies.
This diversity of perspectives and expertise allows us to collaborate rapidly and decisively in our efforts to enhance antibiotic R&D and to effectively examine and exploit previously neglected opportunities. By focusing on some of the most dangerous human pathogens, we will identify novel antimicrobial targets and bacterial vulnerabilities, enhance methods and technologies necessary to overcome the barriers of conventional approaches, and develop a new generation of antimicrobial strategies.
Target Bacteria
Our research targets the microbes posing the greatest threat to human health.
Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus are among the deadliest bacterial pathogens threatening global health, and Brucella melitensis is a significant and endemic issue in several regions worldwide. Our research is guided by a deep understanding of the mechanisms by which these pathogens evade current treatments and our mission is to identify vulnerabilities in their biology and exploit them to develop novel therapies.
Our approach in a nutshell
Our research focuses on three main goals.
First, we aim to develop in-vitro models that replicate key aspects of infection and treatment in patients. These models are benchmarked against healthy human tissue/blood and clinical samples infected with one of the four focus pathogens, and validated with clinically relevant isolates and available data for standard-of-care antibiotics. We enhance the most promising models with biological components such as immune cells, mechanical cues, and specific tissue environments.
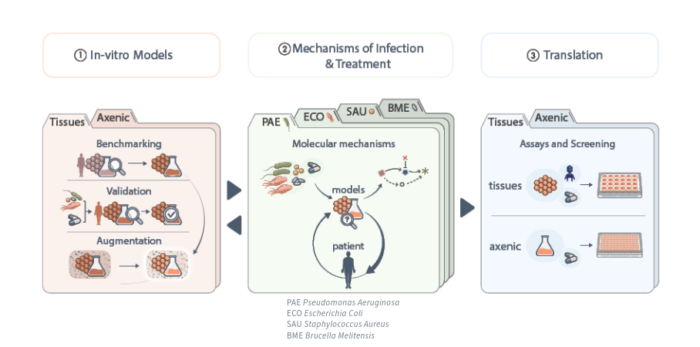
Second, we use these models to discover novel biology, identify overlooked targets, and determine critical mechanisms of antibiotics and new inhibitors under physiologically relevant conditions. This will generate mechanistic hypotheses to be tested in clinics and to address clinically relevant questions using in-vitro models.
Third, based on this mechanistic work and the engineering of improved models for enhanced robustness and throughput, we develop assays for screening of natural products and synthetic compounds, and for the development of non-conventional approaches.
This work is organized into eight work packages, as illustrated in the figure above.